Advanced Copper Electroplating: Techniques and Applications for Industry
Copper electroplating is a well-known process of surface finishing that is extensively used in the contemporary manufacturing industry, and it offers superior corrosion resistance, conductivity and aesthetics. This is a method of electrochemically deposition of a copper layer over a substrate made of metal or any conductive material. Copper electroplating is an important process to manufacturers and engineers used in electronics, automotive, decorative hardware and industrial machinery components. Knowledge on the basics, processes, and uses of advanced copper electroplating enables companies to maximize their manufacturing, enhance the performance of their parts, and increase the life of the components.
What Is Copper Electroplating?
Copper electroplating is the application of a thin copper coat on a conductive surface through the utilization of electrical current in a copper bearing solution. The piece of work functions as the cathode, and the copper ions of the electrolyte are attracted by it and deposited as a uniform metallic layer. This can be used to improve the characteristics of the substrate, such as corrosion resistance, electrical conductivity, and surface fineness. Electroplating necessitates paying close attention to current density, composition of the bath, temperature and time in order to have a regular result on all surfaces.

Materials Suitable for Copper Electroplating
Copper can be plated over a broad spectrum of conductive surfaces such as steel, aluminum, brass as well as nickel-coated surfaces. All the substrates have to be prepared steps so that they can be properly adhesed i.e. cleaning, degreasing and applying of the proper undercoat or strike layer. Other materials, such as plastic, can be lined with copper plating as long as a conductive coating is applied on the non-conductive material first. The quality, durability and uniformity of the copper deposit are directly influenced by the choice of substrate and pre-treatment.
The Electroplating Process
Cleaning and surface preparation is usually the first stage in the production of copper electroplating to remove grease, oil and contaminants. This is then carried on with the immersion of an acidic or neutral copper bath with copper sulfate and sulfuric acid with temperature regulation being maintained to ensure the ion activity. A current of electricity is applied between the cathode and anode, and copper ions will move towards the workpiece. Lastly, the plated part can be rinsed, dried, and post-treated, e.g. polishing or passivated, to improve the quality of the surface and its resistance to corrosion.

Pivotal Factors That Influence the Quality of Copper Plating
There are a number of critical parameters that determine copper electroplating performance and quality. Current density regulates the rate of deposition and uniformity of the thickness whereas temperature of the bath affects the ion mobility and plating efficiency. The electrolyte chemical composition, including additives and level of pH, assists in improving brightness, ductile and leveling copper layer. Adequate agitation will evenly distribute the ions and eliminate flaws like pitting, roughness or uneven cross-sectional thickness.
Varieties of Copper Electroplating Processes
Copper electroplating also exists in several variants based on the need of the application:
Acid Copper Plating: This is a popular method of electronics and PCB manufacturing as it is highly conductive and easily plated.
Alkaline Copper Plating: Gives greater deposits and enhanced throwing capacity on complex geometries or on plating complex surfaces.
Pulse Electroplating: Is based on pulsed current to regulate the structure of deposition, refinement of grain, and internal stress.
Barrel Plating: Small parts can be used and high-volume production is also possible so that part of numerous components can be coated efficiently and uniformly.
All methods possess their own benefits, based on required thickness, surface finish and volume of production.

Uses of Copper Electroplating
Copper Electroplating has many application and industries. In electronics, it is vital to the printed circuit boards, connectors and conductive traces because it is a high electrical conductor. Copper coating is used in automobile production to improve corrosion resistance and provide a surface to be later nickel or chrome plated. Copper plating is also used in ornaments in decorative applications (including hardware, jewelry, and metal ornaments). Wear resistance, thermal conductivity and better adhesiveness to receive further coating are some of the advantages of industrial machinery components.
The Benefits of Copper Electroplating
Copper electroplating has several benefits that render it to be a more preferred process in most manufacturing processes. It has great corrosion resistance that gives the life of the components in the hostile environments. The plating increases the electrical conductivity thus suitable in the electronic components and connectors. Copper reserves are also an effective foundation of other metal layers like nickel or gold. Further, copper electroplating enhances the aesthetic appearance and surface smoothness besides providing low cost production in case of high volume production.
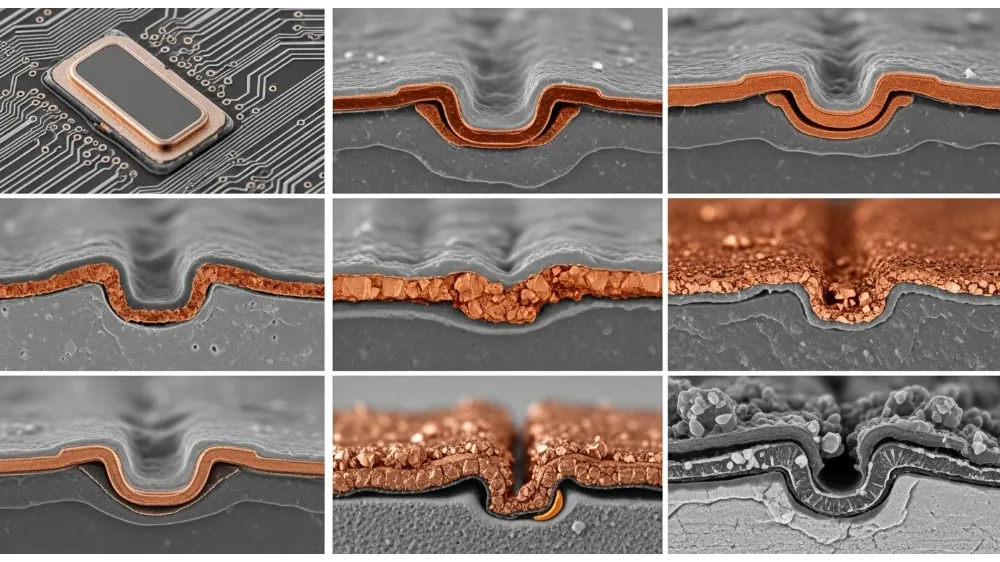
Challenges and Concerns
Copper electroplating, in spite of all its advantages, must be carefully used with consideration to parameters of the process. Thin components may become subjected to internal stress, crack or distort due to over-plating. Due to complicated geometry of the workpiece, uneven deposition can take place necessitating agitation or special anodes. The handling and disposal of used electrolytes should be done in accordance with the environmental policies because copper ions and chemicals may be dangerous. Also, plating baths must be well maintained to prevent contamination and to maintain the same quality through time.
Best Practices for Successful Copper Plating
Best practices are important in order to achieve successful electroplating of copper. This is done by thoroughly cleaning and degreasing substrates to eliminate contaminants. Pay attention to control current density and bath chemistry to attain uniform thickness. Use agitation or circulation to increase the distribution of ions on complicated shapes. Post treatment methods like polishing or passivation should be considered to enhance the look and guard the copper layer. Measurably check the composition of bath and change the depleted chemicals to maintain the process stability and quality of the products.
Environmental and Safety Concerns
Chemicals used in copper electroplating are likely to cause serious effects on the environment and workers of such processes unless dealt with in a proper manner. Protection clothes should be worn by the operators such as gloves, goggles and ventilation masks. Spend electrolytes and wastewater should be treated to eliminate heavy metals and then disposed of. Most of the modern facilities have closed loop systems or recycling methods to limit the chemical waste, environmental damage, as well as conform to safety regulations and still have efficient production.
Future Prospects of Copper Electroplating
The development of copper electroplating is aimed at long-term, accuracy, and automation. Greener plating baths using less toxicity are now being embraced. Consistent thickness and surface quality and minimization of defects are ensured by automated monitoring and control systems. Nanostructured copper coatings and pulse electroplating present an improved mechanical, electrical and thermal performance, and present new opportunities in electronics, aerospace, and enriched manufacturing processes.
Frequently Asked Questions (FAQ)
Q1: What is the purpose of copper electroplating?
Electroplating of metals and copper improves corrosion resistance, electrical conductivity, and appearance of electronic components, automobiles, decorative pieces, and so on.
Q2: Is it possible to plate copper on non-metals?
It is possible to plate non-conductive materials such as plastic provided that a conductive coating is applied.
Q3: What is the average copper plating thickness?
The plating thickness of copper can be a few micron in electronics to a number of hundreds of microns in heavy-duty industrial plating.
Q4: What is the difference between acid and alkaline copper plating?
Precise and smooth electronics Acid copper is accurate and smooth; alkaline copper is superior in the throwing power and thicker deposits.
Q5: What is the impact of copper electroplating on the performance of tools or components?
It enhances electrical conductivity, corrosion resistance, wear resistance, and the smooth surface on which further coating can be applied.
Q6: Are there environmental issues of copper electroplating?
Yes, handling of electrolytes, treatment of chemical waste, and safety precautions should be taken seriously in order to reduce the environmental impact.
Q7: Is copper electroplating automatable?
Yes, industrial quality and efficiency in high-volume production is usually supported by robotic systems and automated monitoring in modern facilities.
Conclusion
Higher copper electroplating is an important procedure in contemporary production, which has superiority of corrosion, electricity and decoration. Manufacturers can obtain quality, durable, and precision metal coating by learning about the correct methods, process parameters, and interconnection between the materials. The correct control, precautionary measures, and care of environmental laws are all that make the production sustainable and reliable, and thus the copper electroplating is essential in electronics, automobile, industrial and decoration.

